Does wearing loupes make your curing light more hazardous to your eyes?
A study of the risks of eye damage clinicians face when using curing lights while wearing dental loupes.
More than 146 million resin-based sealants and restorations are placed annually in the U.S.1 Increased use of resin-based restorative materials also means the increased use of the light-curing unit (LCU) for photopolymerization.
Most LCUs emit in the 400 to 500 nm wavelength ranges depending on the light source emission spectrum, which is vital for a reaction to occur in the light-cure resin restorative materials.2-4 The spectral requirements of the resin-based composite (RBC) materials and the spectral emission of the LCU should correspond to ensure optimum polymerization of restorative dental materials.5,6
One photoinitiator frequently used in resin-based restorative materials is camphorquinone (CQ), which is activated by high-intensity blue light.7,8 LED LCUs correspond with the wavelength necessary to activate the CQ with an ideal narrow blue spectral emission for photoinitiation. Some resin and bonding systems use other photoinitiators in translucent and lighter shade restorative materials.4,9-11
LED LCUs are often the curing unit of choice because of the unit’s high curing efficiency, long life, low energy consumption, and device convenience and ease of use.5 The LED LCU narrow spectral emission is better centered on the peak of maximum absorption of the photoinitiators.5,8,12-14 Although the LCU operates at a blue light spectrum to most effectively cause photoinitiation of RBC materials, it is of an intensity and wavelength that may cause ocular damage.15-17
Related reading: Light curing: What you can't afford to be in the dark about
Blue light hazard
Retinal blue light sensitivity has been identified in the literature for decades. Dental operator’s eyes are at risk from acute and cumulative effects of blue light from inadvertent direct viewing and back reflectance of the light. Retinal damage increases in response to higher energy, short wavelength of the visible spectrum (400 to 500 nm)19,20 which is the spectrum required for photoinitiation.
“Blue-light hazard” (BLH) as identified in the literature is the potential for retinal injury due to high-energy short wavelength light 20 and is greatest at 440 nm.15,19,21 Blue light from LCUs may be particularly damaging because of the frequency and duration of use by the dental operator.
Blue light is transmitted through the ocular media of the eye and absorbed by the retina8,21,22 and high levels may cause immediate and irreversible retinal burning while prolonged exposure to low levels may result in accelerated retinal aging and degeneration.8,21 The chronic injury from low-level blue light exposure is thought to contribute to retinal aging and can accelerate age-related macular degeneration (ARMD).23,24
The Principle of Conservation of Radiance states that the source of radiance and retinal irradiance cannot be increased with the use of optical aids, however, the use of optical aids such as magnifying loupes, increases the image size which may increase the risk of retinal hazard.26 There is little data in the literature regarding the use of loupes in dentistry and their effect for potential increased risk of blue light exposure, so this study aimed to determine whether varying the type of and magnification of loupes would affect the amount of LCU blue light exposure.
Trending article: Are dental loupes improving or worsening your neck health?
Study set up
A VALO blue-light emitting LCU from Ultradent was selected for the experiment. Absolute irradiance from 422 to 525 nm was measured using an Ocean Optics Flame Spectrometer and Ocean View’s software. A cosine corrector optical diffuser with a 3.9 mm diameter was used to collect signal from 180° field of view. An external National Institute of Standards and Technology (NIST) standard light source was used to calibrate the spectrometer at the beginning of the experiment. The LCU and the spectrometer were rigidly mounted on an optical bench (Fig. 1).
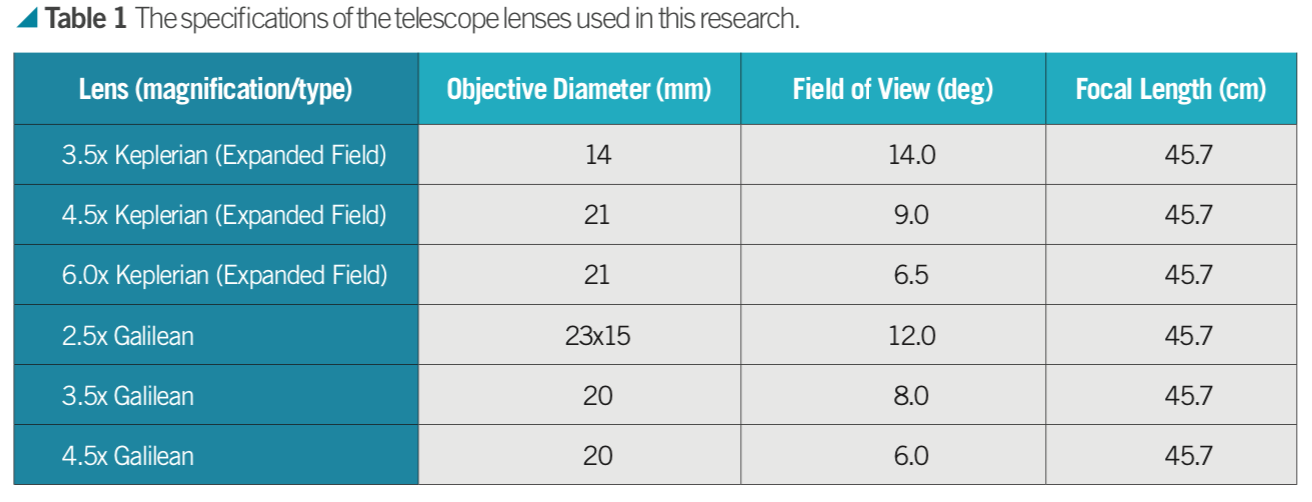
Two different types of telescope lenses were used: Keplerian (expanded field) and Galilean. Lenses were obtained from Designs for Vision, Inc® and for each type of telescope, three different magnifications were used (Table 1).

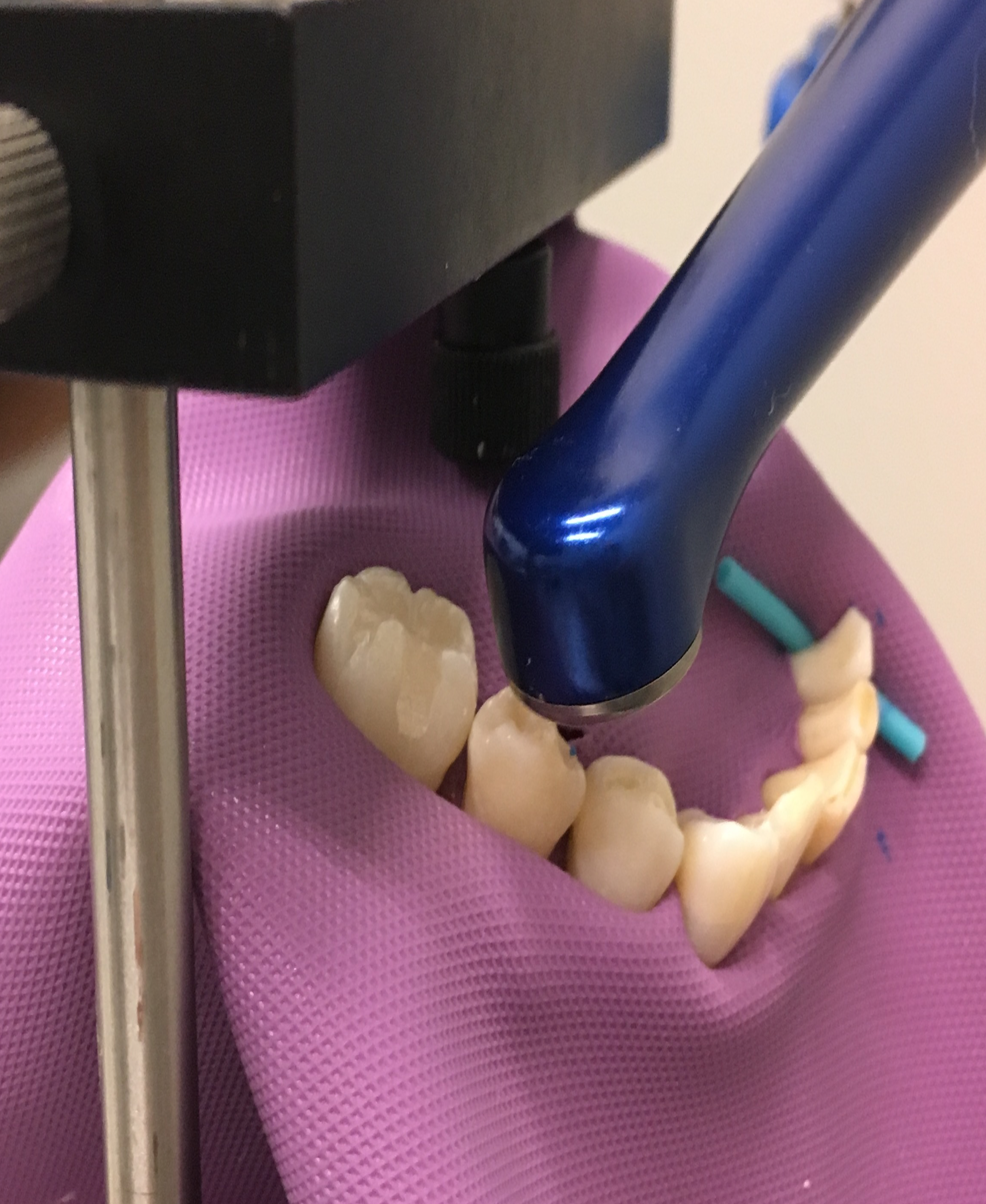
Fig. 1: Spectrometer positioned 13 mm behind lens to simulate the position of the eye exposed to blue light. Fig. 2: Image of configuration set up to measure reflected blue light.
In order to measure the effects of viewing the LCU through magnification loupes, the loupes were positioned 45.7 cm away from the teeth and the LCU. The spectrometer was placed 13 mm behind the loupes to simulate the work- ing distance behind the mounted loupe lenses and the eye in a clinical situation. A sub-analysis with the spectrometer directly in contact with the loupes at a variety of distances up to 25 mm revealed this placement was critical, in particular for the expanded field design.
Image of configuration set up to measure the possible of direct exposure while the curing light is positioned behind the upper incisors.
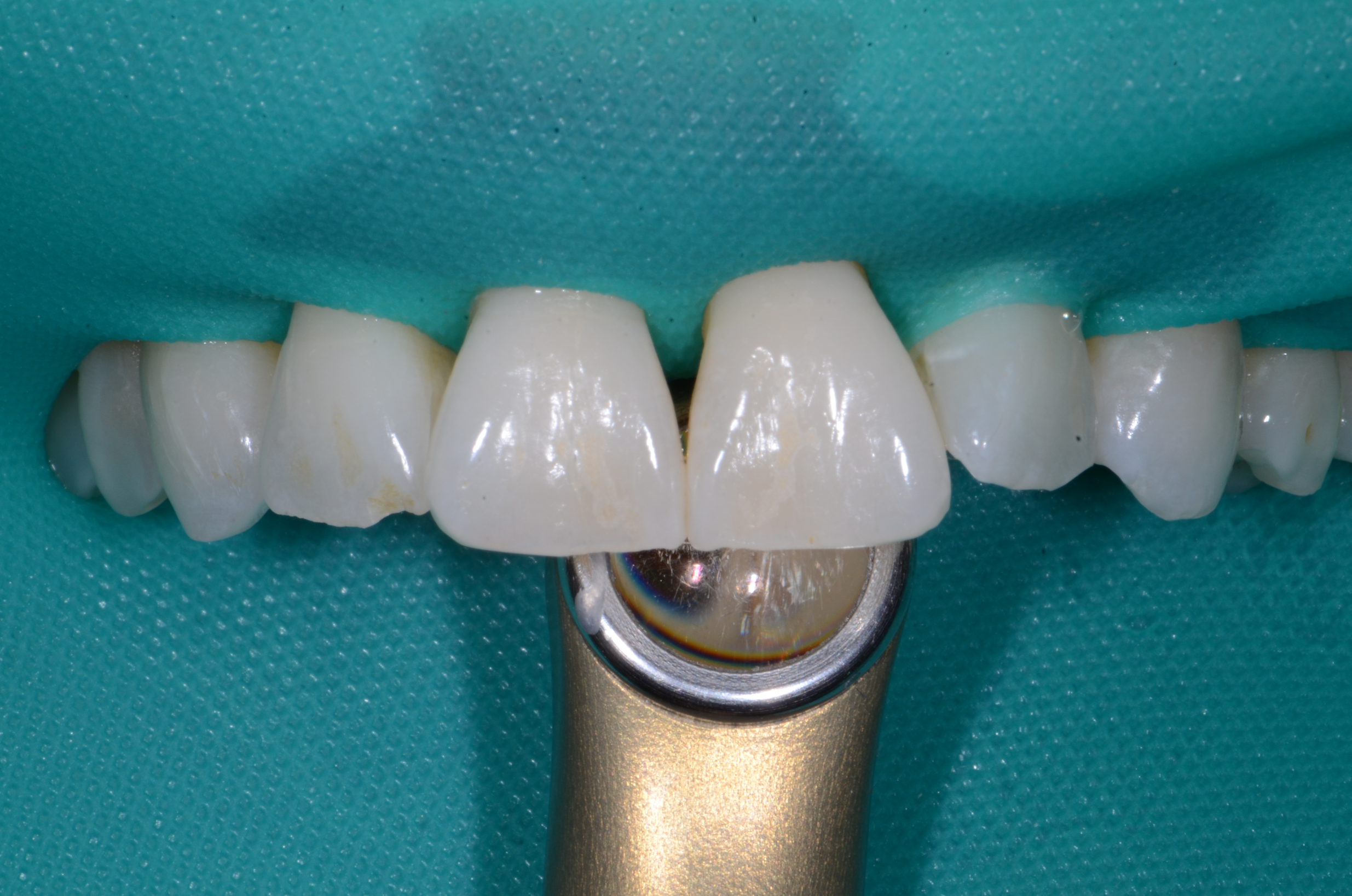
The study was designed to mimic a blue light exposure with a LCU similar to a clinical experience. For the first configuration, measurements were taken five times for each of the lenses with the LCU directed toward natural teeth to evaluate reflected blue light (Fig. 2). Additional measurements were taken for a second configuration with the curing light positioned behind the lingual surfaces of natural maxillary incisors to simulate accidental direct exposure that may occur in that scenario (Fig. 3).
Trending article: How to get the most from your loupes
Study outcomes
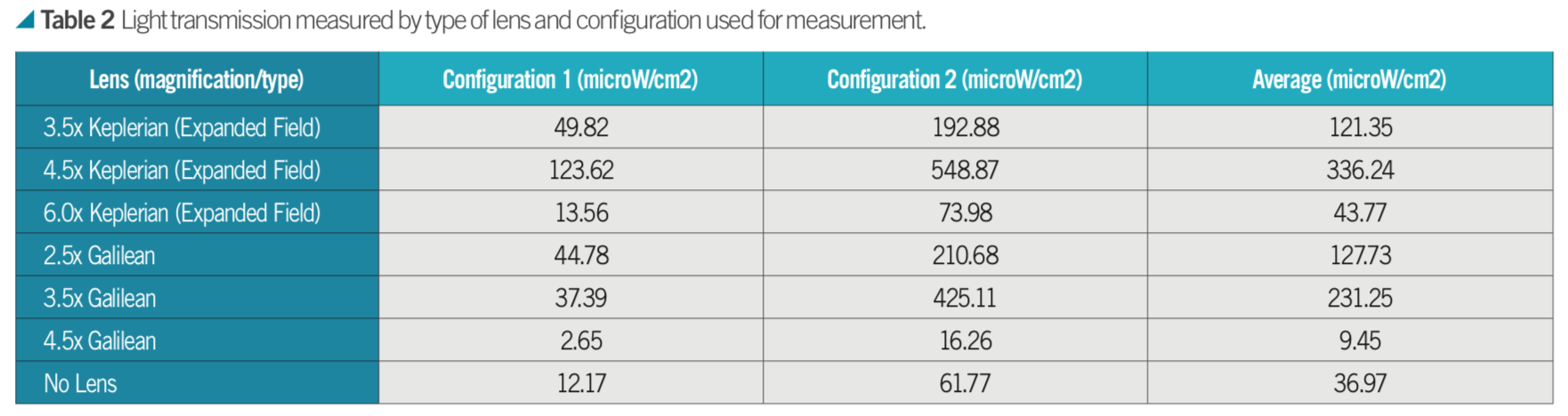
We found that the amount of integrated irradiance measured by the spectrometer through the loupes varied considerably with configuration and type of loupes. There was no clear trend that a larger or smaller magnification resulted in more light transmission with the most light being transmitted by the 4.5x Keplerian lens and the least light by the 4.5x Galilean lens. More light was observed at the corneal plane for all lenses except for 4.5x Galilean (Table 2).
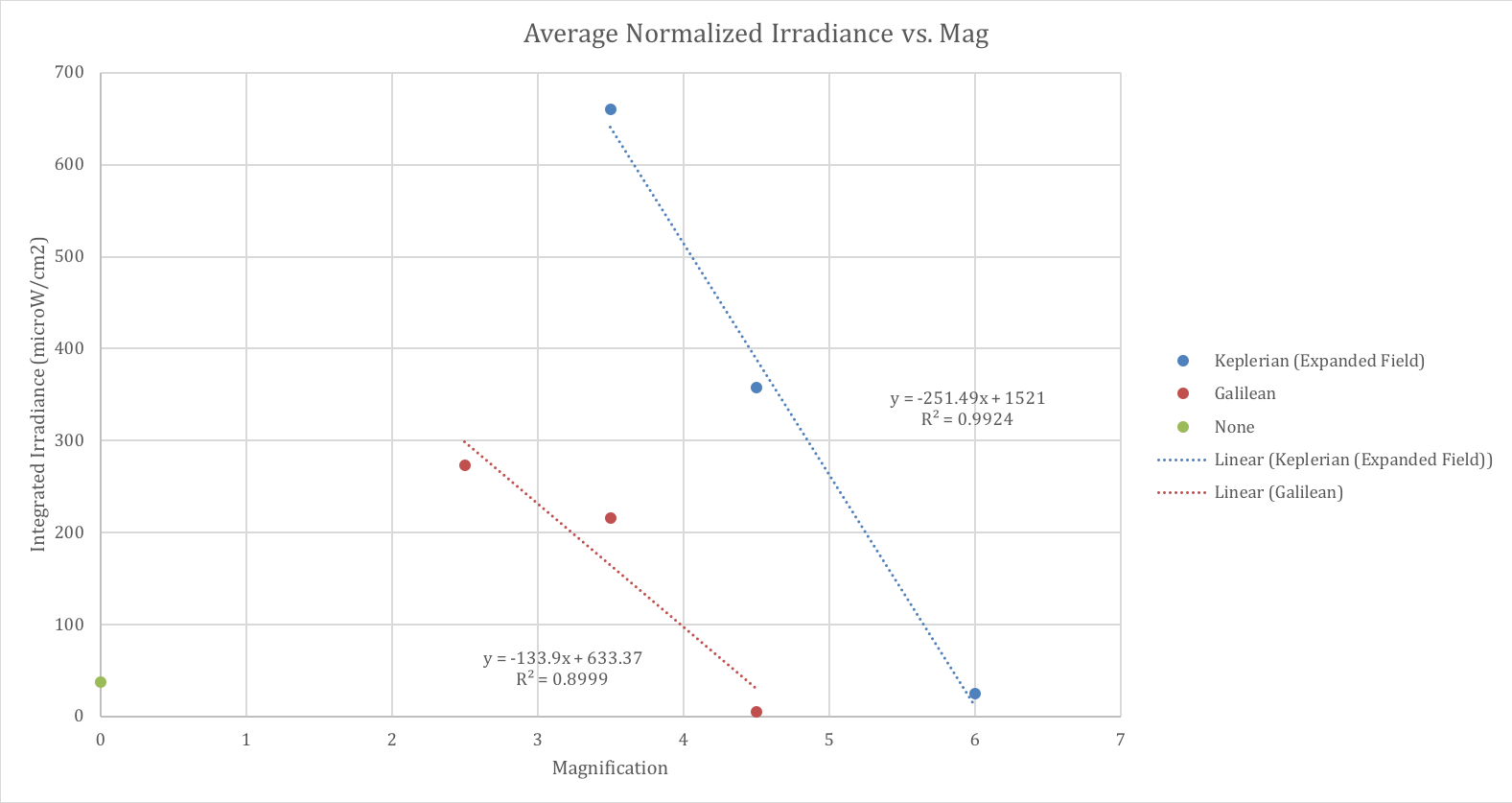
To further understand the trends with telescope type and magnification, we applied two normalization factors. The first was to account for the area of the objective lens because larger collecting optics will result in more light entering the system. The second factor accounted for the relative solid angle subtended by the lens as each had a different field of view. A lens area factor and a solid angle factor, which are intended to normalize the lenses to an average objective diameter and average field of view, were calculated. When these two correction factors were applied to the raw data, clear trends were observed for each telescope type (Fig. 4).
Trending article: 6 important consideration when deciding on loupes
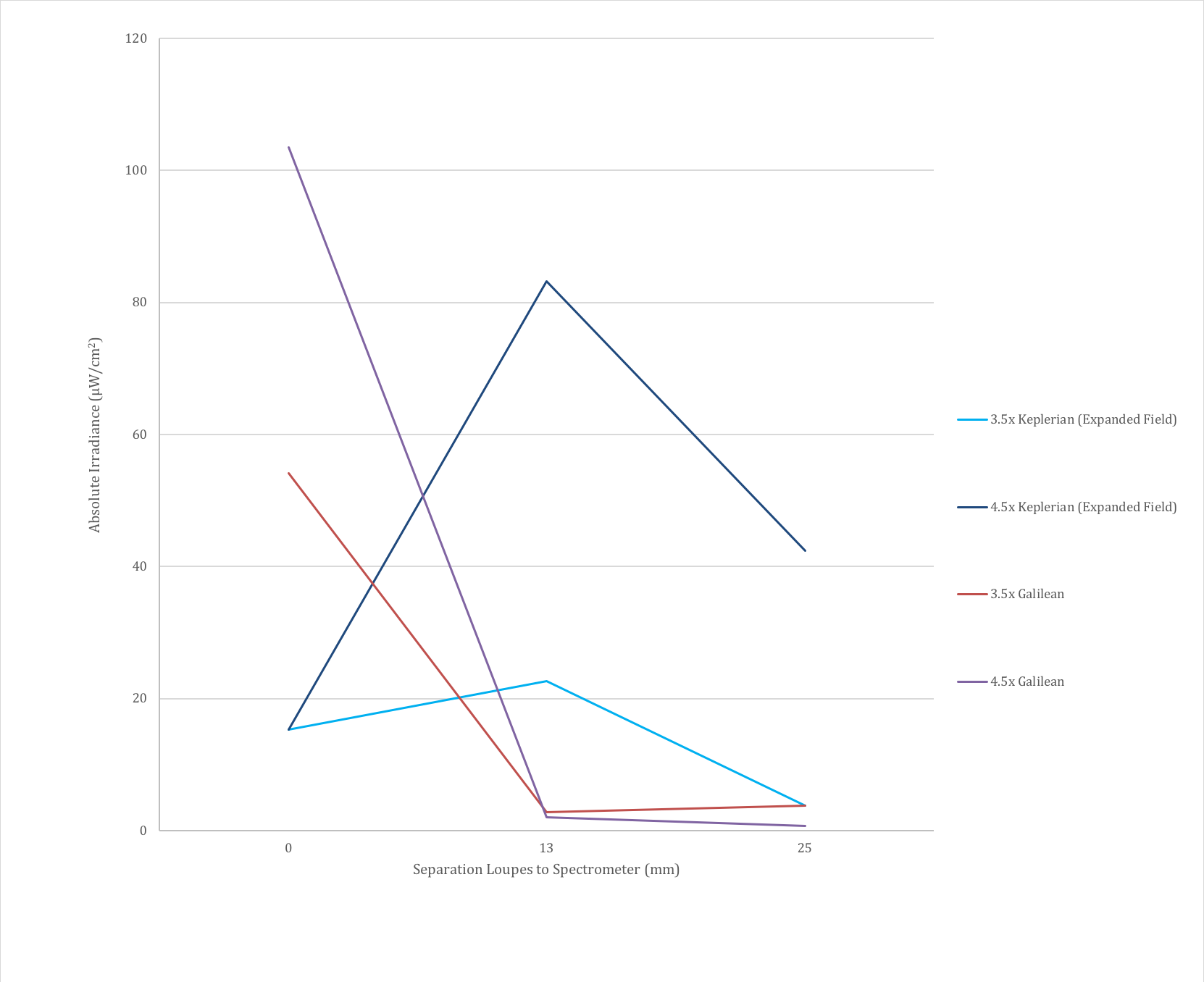
The distance to the telescope matters and results in drastically different results for the Keplerian telescopes compared with the Galilean telescopes. To study this, the absolute irradiance was measured when the spectrometer was in contact with the loupe and at distances of 13 mm and 25 mm behind the loupes. The data indicated there was a significant difference between telescopes, with the Keplerian telescopes increasing in irradiance at a distance of 13 mm com- pared with contact and one inch and the Galilean telescopes abruptly decreasing at a distance of 13 mm and remaining relatively stable in the transition from 13 mm to 25 mm.
The difference is at least partially explained by a significant difference in optical design between the two telescopes, which results in an external exit pupil for the Keplerian design around 13 mm behind the ocular lens, resulting in additional light being measured by the spectrometer. This significant difference means the two telescope designs should be considered separately (Fig. 5).
Related reading: Putting curing lights in the spotlight
Fig. 4: Average Normalized Irradiance vs. Magnification. The integrated irradiance was normalized to account for the size of the telescope objective (entrance pupil) and the field of view.
What this means
Magnification lenses are a vital part of a dental armamentarium. The literature posits the benefits of increased visual acuity, increased productivity, as well as the improved quality of dental care and reduced fatigue and injury.27-31 It is reported that prism loupes have improved magnification, longer working distance, a larger field of view as well as a wider depth of focus.32
Exposure to the risk of blue-light from an LED source can occur in two ways: either from looking at an irradiated scene or directly viewing the light source. During dental procedures, the operator may be directly exposed to a blue light source such as when working on the anterior teeth and a portion of the light-curing tip is not covered by the teeth and the eyes are directly exposed. More commonly, blue light exposure occurs during pho- to polymerization either as a reflected or transmitted source (Fig. 2).
A study of more than 748 dentists reported that almost one-third of dentists used what would be considered as inadequate eye protection against blue light exposure during dental procedures.35 Almost 20 percent of study participants used a LCU mounted shield, while approximately 8 percent looked away from the light.
More on curing lights: Is your curing light 'bright' enough?
Fig. 5: Absolute irradiance measurements with increasing separation between the back of the telescope and the spectrometer for the two magnifications in common between the Keplerian and Galilean designs. There is a clear difference between designs here, with the Galilean designs quickly decreasing in absolute irradiance whereas the Keplerian designs first increase and then decrease with increasing distance behind the loupes. The Keplerian design has a real exit pupil, which can account for this increase in absolute irradiance.
Eye aversion to bright light is a natural response that may limit the exposure time to less than 0.25 seconds.7,8,33,36 Unfortunately, blue light from an LCU may not induce the same protective response and thereby increase blue light exposure time due to a delayed pupil constriction response. Furthermore, the operator is trained to watch to make sure they align the curing light to the tooth being treated to assure ultimate cure of the restorative material. Clinicians who avert their eyes run the risk of not correctly aligning the curing light to the restoration, possibly leading to inadequate photopolymerization of the restorative material.
Light wavelengths of under 400 nm are absorbed by the lens of the eye and do not reach the retina. Blue spectrum radiation can reach the retina and in the young eye ocular transmittance can be close to 90 percent at 450 nm.33 Blue light damage may be resultant to a photochemical process causing injury to the pigmented epithelium and choroid of the retina.23,37,38 The extent of retinal injury is dependent on the efficiency of the antioxidant system.20,39 Dental professionals who have undergone cataract removal would be more susceptible to damage from blue-light exposure.43
Several studies identified the cumulative character of light damage to the eyes.45,46 Interestingly, a study found that three and four exposures of five minutes duration followed by a one-hour dark interval led to significantly more damage.37 Such light dose fractionation can elicit a more harmful effect than the same dose of light without interruption. This may be of particular concern for dental personnel because of the repetitive use of the LCU throughout the day.
Hot read: Tips and tricks for creating long-lasting restorations
Based on the results of this study, the use of expanded field Keplerian dental loupes 13 mm from the magnification lens may increase the risk of blue light exposure to the dental operator (Fig. 5).
In one study based on an eight-hour day, the maximum permissible cumulative exposure time as calculated according to the guidelines as set by the American Conference of Governmental Industrial Hygienists (ACGIH) would be achieved in approximately 11 minutes. The use of loupes was found to increase the maximum exposure time to the pupil up to 28 minutes.36
Dentists may spend 240 hours per year curing dental restorations.48 One study of dentists in the clinical setting reported 57.5 percent of their working day was spent placing light cured restorations.35
One-fourth of dentists use the shield mounted to the curing light for eye protection.35 However, these shields may not be beneficial as the only form of protection as they are too small to protect against light spreading in all directions. Blue-light filtering spectacles cause a reduction in the transmission of light below 500 nm to less than one percent.51 Another option for safety is a paddle-designed filter to protect the eyes of clinicians.52
Trending article: Enhanced technologies for faster, easier dental treatments
Conclusion
Blue-light hazard is the potential for retinal injury due to high-energy short wavelength light and is greatest at 440 nm. Most LCUs emit in the 350-450 nm wavelength range and the absolute irradiance received by the eye may be greater with expanded field telescopes. Lack of eye movement and focus while using magnification may increase the potentially damaging effects of greater radiant exposure to the retina.
Whether or not they are wearing loupes at the time, dental professionals should protect their eyes when using LCUs. Protective spectacles or paddles designed to filter out the harmful wavelengths should be used.
Trending article: 6 ways bulk fills are changing restorative dentistry
Special thanks to Midwestern University – Arizona for supporting our research and Designs for Vision, Inc.® for providing telescopic lenses used in the study. Also thanks to Allen Tang who was a student of Midwestern University at the time of the study for his assistance.
References
- American Dental Association ADA 2005-06 Survey of Dental Services Rendered; [cited 2018 Oct 3] Available from: https://www.ada.org/
- Roll EMB, Jacobsen N, Hensten-Pettersen A. Health hazards associated with curing light in the dental clinic. Clin Oral Investig 2004 Sep;8(3):113-117.
- Uhl A, Sigusch BW, Jandt KD. Second generation LEDs for the polymerization of oral biomaterials. Dent Mater 2004 Jan;20(1):80-87.
- Park YJ, Chae KH, Rawls HR. Development of a new photoinitiation system for dental light-cure composite resins. Dent Mater 1999 Mar;15(2):120-127.
- Jandt K, Mills R. A Brief History of LED Polymerization. Dental Materials 2013 Jun;29:605-617.
- Price RB, Fahey J, Felix CM. Knoop microhardness mapping used to compare the efficacy of LED, QTH and PAC curing lights. Oper Dent 2010 Jan;35(1):58-68.
- Srivastava SC, Verma K, Azam A, Sharma M. Blue Light Hazard from Photopolymerization Unit: Literature Review. Asian Journal of Oral Health & Allied Sciences 2014;4(1):11-15.
- Price RB, Shortall AC, Palin WM. Contemporary issues in light curing. Oper Dent 2014 Jan/Feb;39(1):4-14.
- Ogunyinka A, Palin WM, Shortall AC, Marquis PM. Photoinitiation chemistry affects light transmission and degree of conversion of curing experimental dental resin composites. Dent Mater 2007 Jul;23(7):807-813.
- Neumann M, Miranda Jr. W, Schmidt C, Rueggeberg F, Correa I. Molar Extraction coefficients and the photon absorption efficiency of dental photoinitiators and light curing units. Journal of Dentistry 2016 Mar;33(6):525-532.
- Ilie N, Hickel R. Can CQ be completely replaced by alternative initiators in dental adhesives? Dent Mater J 2008 Mar;27(2):221-228.
- Rueggeberg FA. State-of-the-art: dental photocuring--a review. Dent Mater 2011 Jan;27(1):39-52.
- Price R, Labrie D, Rueggeberg F, Felix C. Irradiance differences in the violet (405 nm) and blue (460 nm) spectral ranges among dental light-curing units. J Esthet Restor Dent 2010 Dec;22(6):363-377.
- Leprince J, Devaux J, Mullier T, Vreven J, Leloup G. Pulpal-temperature rise and polymerization efficiency of LED curing lights. Oper Dent 2010 Mar/Apr;35(2):220-230.
- Labrie D, Moe J, Price RB, Young ME, Felix CM. Evaluation of ocular hazards from 4 types of curing lights. J Can Dent Assoc 2011Oct;77:b116.
- McCusker N, Lee SM, Robinson S, Patel N, Sandy JR, Ireland AJ. Light curing in orthodontics; should we be concerned? Dent Mater 2013 Jun;29(6):e85-90.
- Bruzell Roll EM, Jacobsen N, Hensten-Pettersen A. Health hazards associated with curing light in the dental clinic. Clin Oral Investig 2004 Sep;8(3):113-117.
- Ham WT, Mueller HA, Sliney DH. Retinal sensitivity to damage from short wavelength light. Nature 1976 Sep;260(5547):153-155.
- Algvere PV, Marshall J, Seregard S. Age-related maculopathy and the impact of blue light hazard. Acta Ophthalmol Scand 2006 Feb;84(1):4-15.
- Stamatacos C, Harrison JL. The possible ocular hazards of LED dental illumination applications. J Tenn Dent Assoc 2013 Apr;93(2):25-9; quiz 30-1.
- Rueggeberg F, Giannini M, Augusto C, Arrais G, Bengt R, Price T. Light curing in dentistry and clinical implications: A literature review. Braz Oral Res 2017Aug;31(e61):64-91.
- Melton R. The lowdown on blue light: Good vs. bad and it connection to AMD. Review of Optometry [Internet]. 2014 Feb;1-3; [cited 2018 Oct] Available from: https://www.reviewsce.com
- Ham WT, Ruffolo JJ, Mueller HA, Clarke AM, Moon ME. Histologic analysis of photochemical lesions produced in rhesus retina by short-wave-length light. Invest Ophthalmol Vis Sci 1978 Oct;17(10):1029-103535.
- International Commission on Non-Ionizing Radiation Protection. Guidelines on limits of exposure to broad-band incoherent optical radiation (0.38 to 3 microM). Health Phys 1997 Sep;73(3):539-554.
- Ham WT, Ruffolo JJ, Mueller HA, Guerry D. The nature of retinal radiation damage: dependence on wavelength, power level and exposure time. Vision Res 1980:20(12):1105-1111.
- ICNIRP statement on light-emitting diodes (LEDS) and laser diodes: implications for hazard assessment. International Commission on Non-Ionizing Radiation Protection. Health Phys 2000 Jun;78(6):744-752.
- Perrin P, Ramseyer ST, Eichenberger M, Lussi A. Visual acuity of dentists in their respective clinical conditions. Clin Oral Investig 2014 Dec;18(9):2055-2058.
- Eichenberger M, Perrin P, Neuhaus KW, Bringolf U, Lussi A. Visual acuity of dentists under simulated clinical conditions. Clin Oral Investig 2013 Apr;17(3):725-729.
- Farook SA, Stokes RJ, Davis AK, Sneddon K, Collyer J. Use of dental loupes among dental trainers and trainees in the UK. J Investig Clin Dent 2013 May;4(2):120-123.
- Meraner M, Nase JB. Magnification in dental practice and education: experience and attitudes of a dental school faculty. J Dent Educ 2008Jun;72(6):698-706.
- Perrin P, Eichenberger M, Neuhaus KW, Lussi A. Visual acuity and magnification devices in dentistry. Swiss Dent J 2016;126(3):222-235.
- Shanelec DA. Optical principles of loupes. J Calif Dent Assoc 1992 Nov;20(11):25-32.
- Mathew J, Manuja N, Nair N, james B, Syriac G. Ocular hazards from use of light-emitting diodes in dental operatory. Journal of Indian Academy of Dental Specialists Researchers 2017Aug;4:28-31.
- Mathôt S, Van der Stigchel S. New light on the mind's eye:The pupillary light response as active vision. Curr Dir Psychol Sci 2015 Oct;24(5):374-378.
- Kopperud SE, Rukke HV, Kopperud HM, Bruzell EM. Light curing procedures - performance, knowledge level and safety awareness among dentists. J Dent 2017 Mar;58:67-73.
- Price RB, Labrie D, Bruaell E, Sliney D, Strassler H. The dental curing light: A potential health risk. J Occup and Environ Hyg 2016 Aug;13(8):639-646.
- Noell W, Walker V, Kang BS, Berman S. Retinal damage by light in rats. Invest Opthamol 1966 Oct;5(5):450-473.
- Wu J, Seregard S, Algvere PV. Photochemical damage of the retina. Surv Ophthalmol 2006 Sep-Oct;51(5):461-481.
- Organisciak D, Winkler B. Retinal light damage: practical and theoretical considerations. Prog Retinal Eye Res, 1994;1-29.
- Behar-Cohen F, Martinsons C, Viénot F, Zissis G, Barlier-Salsi A, Cesarini JP, Enouf O, Garcia M, Picaud S, Attia D. Light-emitting diodes (LED) for domestic lighting: any risks for the eye? Prog Retin Eye Res 2011 Jul;30(4):239-257.
- Ham WT, Mueller HA, Ruffolo JJ, Guerry RK, Clarke AM. Ocular effects of GaAs lasers and near infrared radiation. Appl Opt 1984 Jul;23(13):2181-2186.
- Hunter JJ, Morgan JI, Merigan WH, Sliney DH, Sparrow JR, Williams DR. The susceptibility of the retina to photochemical damage from visible light. Prog Retin Eye Res 2012:31(1):28-42.
- Cuthbertson FM, Peirson SN, Wulff K, Foster RG, Downes SM. Blue light-filtering intraocular lenses: review of potential benefits and side effects. J Cataract Refract Surg 2009 Jul;35(7):1281-1297.
- Dawe RS, Ibbotson SH. Drug-induced photosensitivity. Dermatol Clin 2014 Jul;32(3):363-368.
- Ham WT, Mueller HA, Ruffolo JJ, Clarke AM. Sensitivity of the retina to radiation damage as a function of wavelength. Photochem Photobiol 1979 Apr;29(4):735-743.
- Lawwill T, Crockett S, Currier G. Retinal damage secondary to chronic light exposure, thresholds and mechanisms. Doc Ophthalmol 1977:44(2):379-402.
- Ness J, Swick H, Stuck B, Lund D, Lund B, Molchany J. Retinal image motion during deliberate fixation: implications to laser safety for long duration viewing. Health Phys 2000 Feb;78(2):131-142.
- Christensen GJ. Curing restorative resin: a significant controversy. J Am Dent Assoc 2000 Jul;131(7):1067-1069.
- Christensen G. Is your LED headlamp damaging your eyes? Clin Rep 2013 Mar;6(3).
- Ide T, Kinugawa Y, Nobae Y, Suzuki T, Tanaka Y, Toda I, Tsubota K. LED light characteristics for surgical shadowless lamps and surgical loupes. Plast Reconstr Surg Glob Open 2015 Dec;3(11):e562.
- Bruzell EM, Johnsen B, Aalerud TN, Christensen T. Evaluation of eye protection filters for use with dental curing and bleaching lamps. J Occup Environ Hyg 2007 Jun;4(6):432-439.
- Soares CJ, Rodrigues MP, Vilela AB, Rizo ER, Ferreira LB, Giannini M, Price RB. Evaluation of eye protection filters used with broad-spectrum and conventional LED curing lights. Braz Dent J 201 Jan/Feb;28(1):9-15.