LeEject 2 Syringe and Needle system receives FDA clearance
The system is said to allow disposal of the needle without recapping and unscrewing.
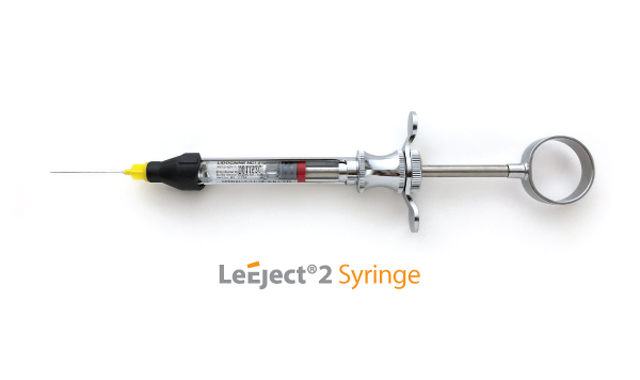
The LeEject 2 Syringe and Needle system has received FDA clearance for sale in the United States. This unique system, protected by a U.S. patent, reportedly differs from conventional dental syringe and needle systems because it allows the loading of the needles from the side portal and disposal of the needle without recapping and unscrewing.
The needle is secured by inserting it from the side and rotating the plastic sheath clockwise. The loading and unloading procedures of anesthetic cartridges are the same as the conventional dental syringe and needle system. The removal of the needle is said to be very simple; by rotating the safeguard counterclockwise and inverting the syringe over a sharps container, and letting the needle fall into it, this eliminates the potential risk of recapping and unscrewing the contaminated needle.
Trending article: 5 reasons you need an infection prevention coordinator
Currently, OSHA prohibits recapping or removal of contaminated needles and other contaminated sharps unless the employer can demonstrate that no alternative is feasible. Because of the lack of alternatives in the market, recapping or needle removal must be accomplished using a mechanical device or a one-handed technique.
The LeEject 2 system is reportedly safe, simple and economical. Dr. Alexander Lee, DDS, who has been overseeing the application process to the FDA for the past six years, wants to concentrate on marketing toward dental schools, hygiene programs and residencies by providing the systems at a reduced cost to them because inexperienced operators are more likely to sustain needle stick injuries.
For more information, visit www.LeEject.com.