The importance of oral cancer screening in the dental practice
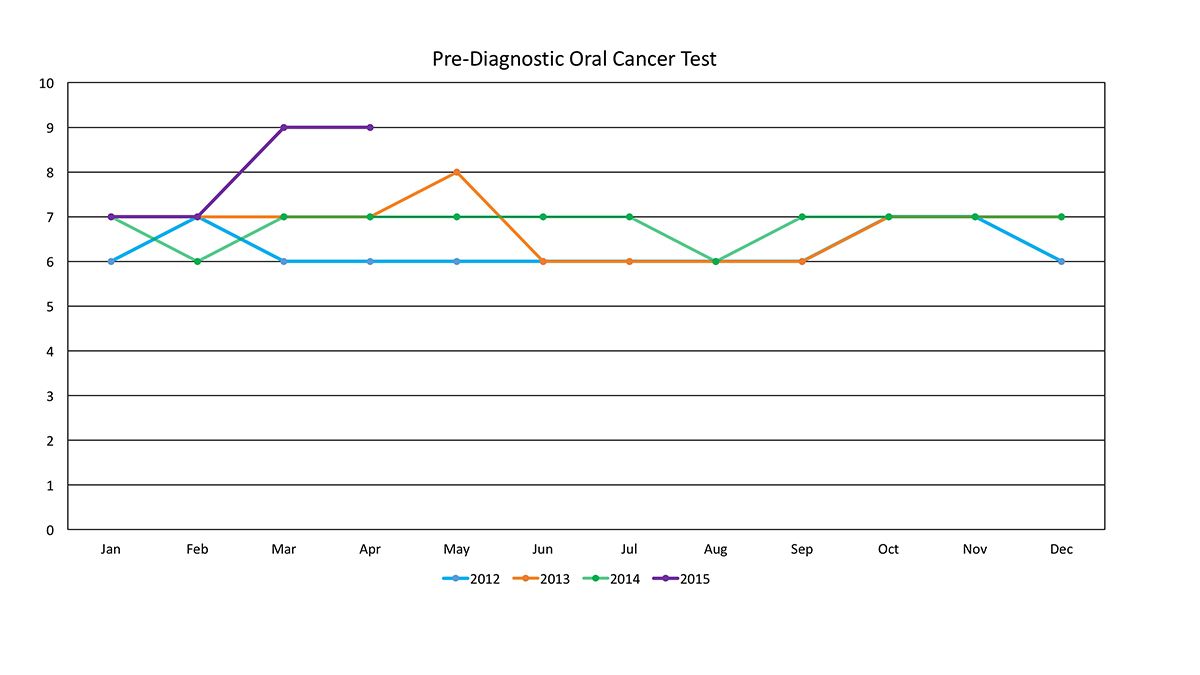
The national average of number of adjunct oral cancer screenings billed under CDT D0431 per clinic has risen sharply. From 2012 to 2014, clinics performed an average of six to seven tests each month.
The dentist’s office is the first line of defense for oral cancer diagnosis.
Approximately 60 percent of the population receives regular dental care1 and these visits are a unique opportunity to look for signs of cancer growth and discuss changes the patient has experienced in his or her mouth.
There are two ways dentists look for oral cancer: The first is a visual examination of the mouth and the second is through the use of adjunctive screening tools such as Velscope and ViziLite. Without vigilance, cancer diagnosis may be delayed which often translates to poorer outcomes and higher mortality rates. Oral cancer that is detected early has an 80 percent five-year survival rate compared to the overall five-year survival rate of 59 percent1.
Sikka Software collects data from thousands of practices across the nation. Data is collected in accordance with privacy laws. This article will discuss trends in technology based screening under CDT D0431, adjunctive pre-diagnostic testing.
Related reading: The top 5 reasons to really think about oral cancer
Oral cancer incidence, signs and symptoms
According to the New York Times, one in 99 people will develop oral cancer in their lifetime. This translates to approximately 45,750 oral and pharyngeal cancers annually with 8,650 deaths2. Cancer is often associated with heavy drinking, use of tobacco products, UV exposure and more recently, HPV163. According to the Oral Cancer Foundation, HPV16-related oral cancers can be particularly challenging to identify as it tends to be in the back of the mouth and patients tend to be younger than the traditional oral cancer patients. An increase in smoking among women has possibility contributed to the move towards equalization in oral cancer gender ratios, although men are twice as likely to get oral cancer than women2.
Oral cancer can be difficult to diagnose as there are generally few physical symptoms in the early stages. Visible physical changes may appear similar to other pathologies, like canker sores, increasing the challenge of diagnosing cancer early. A visual exam of high risk patients has found to reduce mortality 34 percent, however, 25 percent of cancer patients are not considered high risk2, opening up the debate for the place of technology-based screening tools in the clinic.
Related reading: Common misconceptions about oral cancer prevention
Continue to page two for more...
Oral cancer pre-diagnostic testing
The national average of number of adjunct oral cancer screenings billed under CDT D0431 per clinic has risen sharply. From 2012 to 2014, clinics performed an average of six to seven tests each month. In contrast, the number of screenings has rapidly risen to nine tests in the spring of 2015. Although this is a short-term trend, it is far above the previous average. It is possible that this trend is due to an increase in HPV16 related cases, which are difficult to diagnosis visually due to the fact that they are often found in the back of the mouth2.
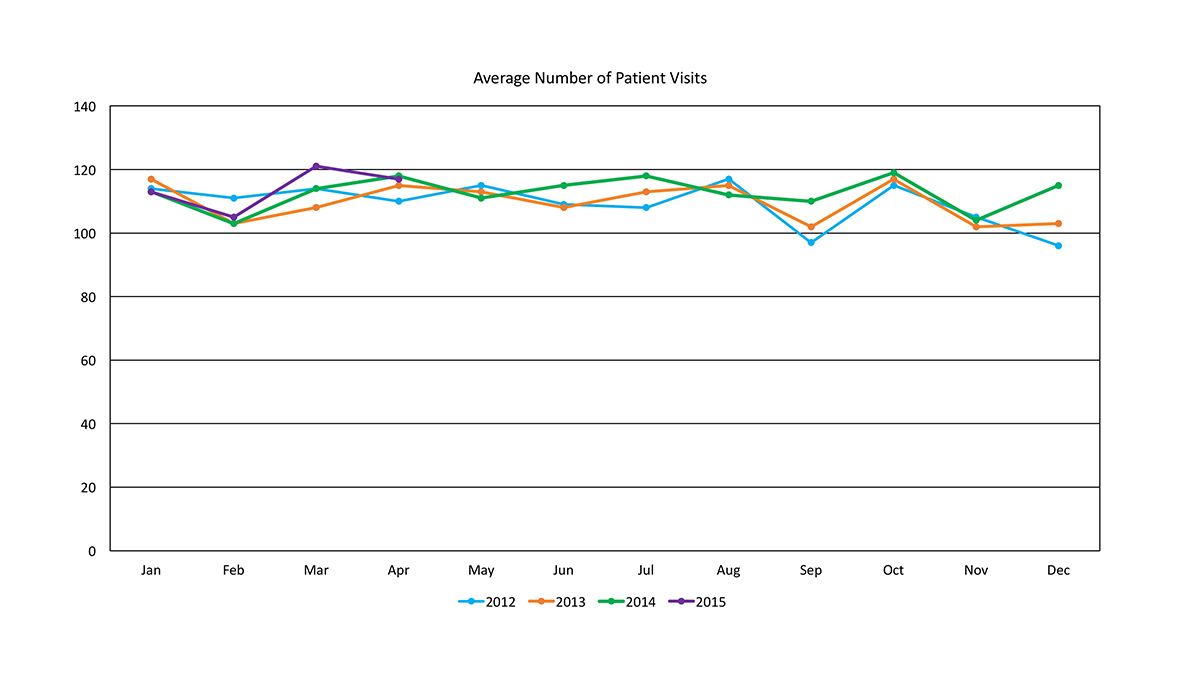
When compared to number of patient visits, the number of oral cancer screenings is small. During the first four months in 2015, the average number of patient visits was 114, while the average number of oral cancer screenings was eight. This indicates that while dentists may be watching for signs of cancer, they are not as frequently using technology and billing the CDT 4D031 code. Dentists may not complete this type of testing because of cost concerns, level of cancer risk to the patient and time constraints.
More on oral cancer: The link between oral sex and oral cancer
Monthly revenue
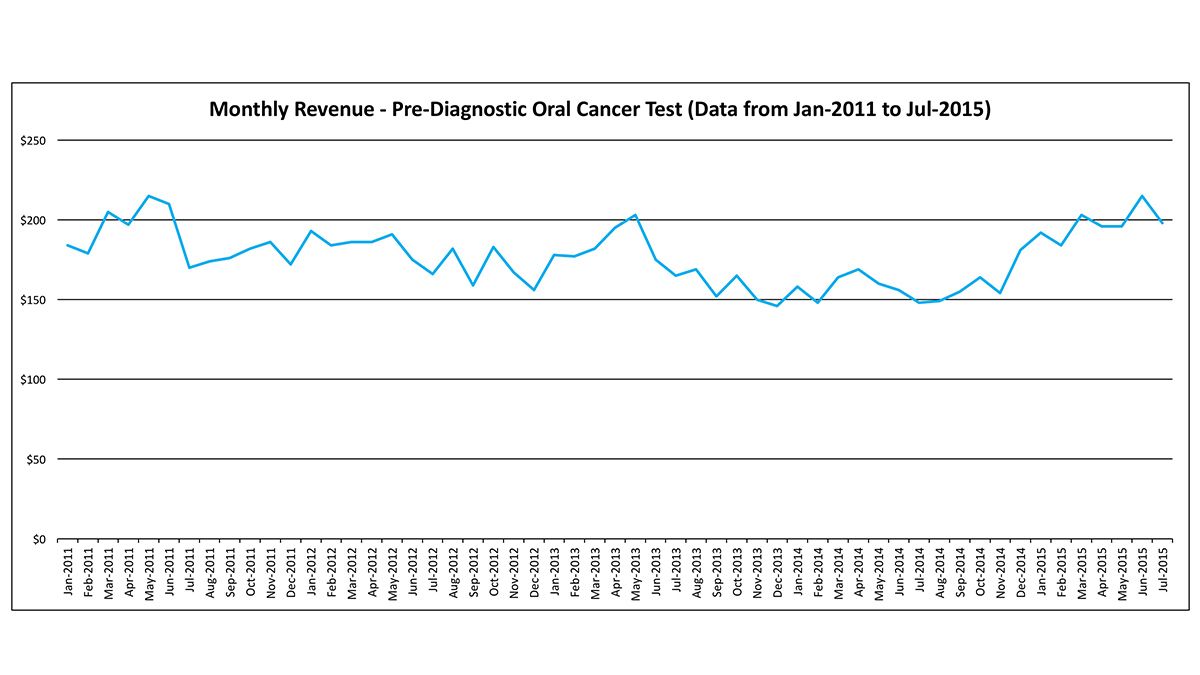
The average monthly revenue generated by a dental office from the oral cancer pre-diagnostics code was around $200, but sloped down to as low as $150 in 2013 and 2014. Revenue rose in 2015 back up to around $200, consistent with the increase in number of screenings performed in clinics.
Conclusion
Dentists are the front line of defense for detecting possible oral cancers. Although oral cancer can be challenging to diagnose in the early stages, dentists are the most likely to note changes in the mouth during regular examinations. Finding these changes are critical as early detection greatly increases the change of five-year survival. The ADA recommends that clinicians remain vigilant for changes to in the mouth that may indicate the presence of cancer. However as the large-scale efficacy of adjunct screening tools remains in question4, there are no current published guidelines for use.
Related reading: Oral cancer awareness survey reveals greater need for education, screening tools
About Sikka Software
Sikka Software provides a platform for small and medium-sized business healthcare apps. The company’s products help healthcare providers and other small businesses optimize their business via a series of easy to use cloud-based applications. With over 32 apps built on Sikka Platform Cloud with over 14,3000 installations, Sikka Software is the leader in the US Dental, Animal Health and Hearing Care markets. The company is now supporting a real time optimization and information network with providers, patients, consultants, manufacturers and financial service provides.
A privately-held company, Sikka Software is headquartered in Milpitas, California with offices in the United States and in India. More information may be found at www.sikkasoftware.com.
About the author
Alitta Boechler is a marketing manager at Sikka Software. She holds a Doctorate of Audiology from the University of Iowa and an MBA from the Jesse H. Jones Graduate School of Business at Rice University. You can contact her at alitta.boechler@sikkasoftware.com or (408) 359-3014. For more information about The Practice Optimizer Company® please visithttps://www.sikkasoft.com/dental/sikka-apps/practice-optimizer-dental/
Sources
1 Tarkan, L. (2009, February 2). New oral cancer tests: crucial or wasteful? The New York Times. Retrieved from http://www.nytimes.com/2009/02/03/health/03cancer.html?_r=1
2 The Oral Cancer Foundations (2015). Oral cancer facts. Retrieved from http://www.oralcancerfoundation.org/facts/
3 British Dental Health Foundation (2015). Facts and figures- mouth cancer action month. Retrieved from http://www.mouthcancer.org/facts-figures/
4 Rethman, M.P, Carpenter, W., Cohen, E.E.W., Esptein, J., Evans, C.A., Flaitz, C.M., … Meyer, D.M. (2010). Evidence-based clinical recommendations regarding screening for oral squamous cell carcinomas. The Journal of the American Dental Association, 141(5), 509-520. Retrieved from http://jada.ada.org/article/S0002-8177(14)61524-5/fulltext#cesec60
American Dental Association (2015). Clinical practice guidelines. Retrieved from http://ebd.ada.org/en/evidence/guidelines