Glidewell Announces Silent Nite Sleep Appliance with Glidewell Hinge
Glidewell Hinge will now be added to the Silent Nite Sleep Appliance, allowing it to be covered under Medicare as well as help patients with obstructive sleep apnea.
Glidewell Announces Sleep Nite Sleep Appliance with Glidewell Hinge
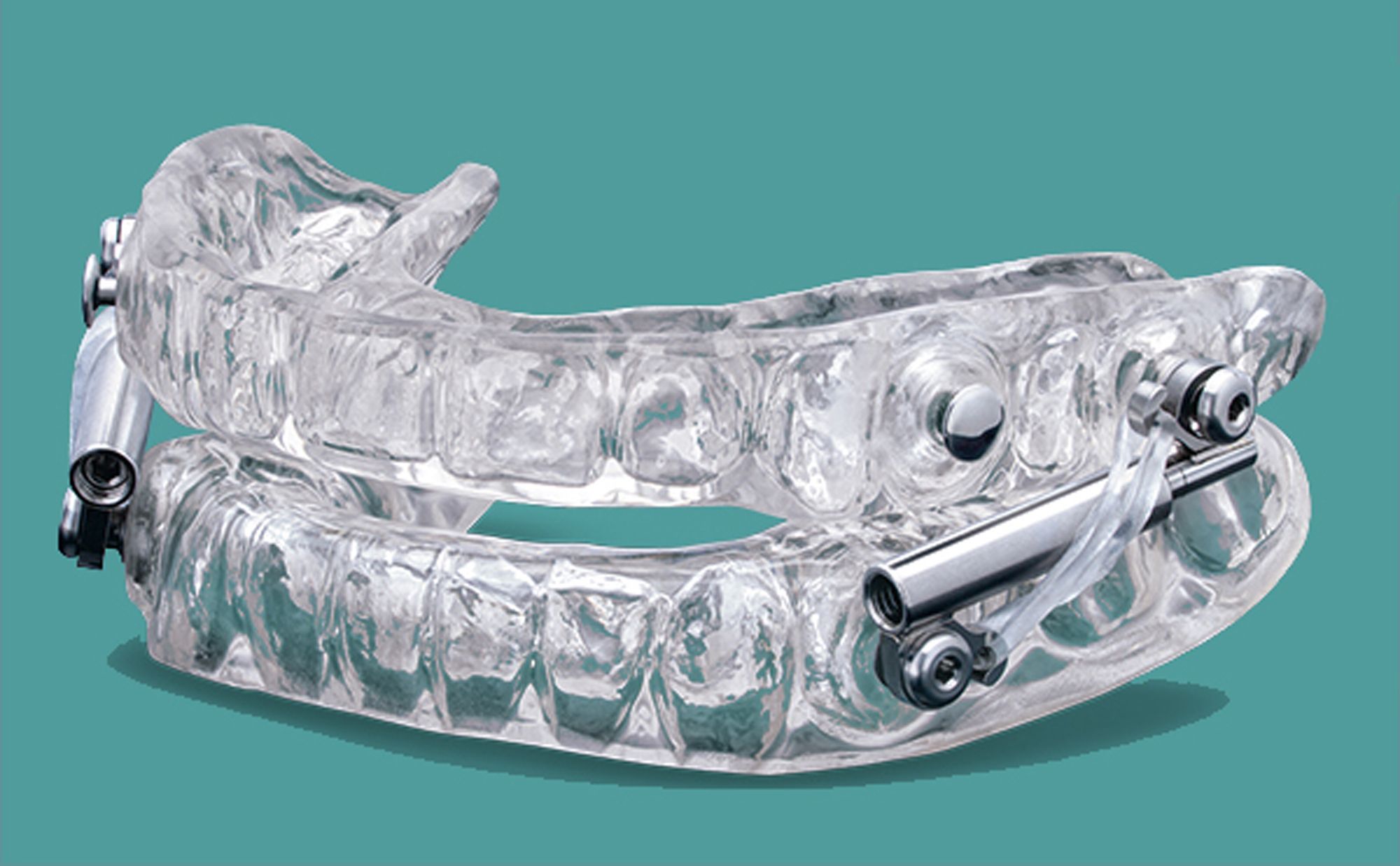
Glidewell has announced the Silent Nite® Sleep Appliance with Glidewell Hinge™. Silent Nite with Glidewell Hinge will combine to treat obstructive sleep apnea (OSA) and snoring. The device will enable patients that use Silent Nite to be covered under their Medicare, according to Glidewell Founder and President Jim Glidewell, CDT.
“I’m pleased to announce the availability of the Silent Nite with Glidewell Hinge,” Glidewell said in a press release from the company. “This device is the culmination of years of innovation from our world-class R&D department, in collaboration with dentists. The new Glidewell Hinge design feature not only expands the use of the Silent Nite to patients with a diagnosed OSA condition regardless of medical insurance carrier, but it also reduces costs to clinicians and improves the ease of prescribing sleep therapy in the general practice.”
Because of the design criteria in place for mandibular advancement devices covered under Medicare, Glidewell research and development designed Hinge to specifically meet these requirements for patients who need it the most. It is made of surgical stainless steel and is micro-adjustable to therapeutic jaw positioning inside or outside of the mouth.
The Hinge device uses a bilateral compression that pushes the jaw into position while also incorporating elastics to soften jaw activity. This is said to encourage closed-mouth nasal breathing without sacrificing patient comfort.