The Evolution of Nonsurgical Periodontal Therapy: Part II
In this 3-part series, we examine the history and evolution of nonsurgical periodontal therapy.
©Alex Mit/stock.adobe.com
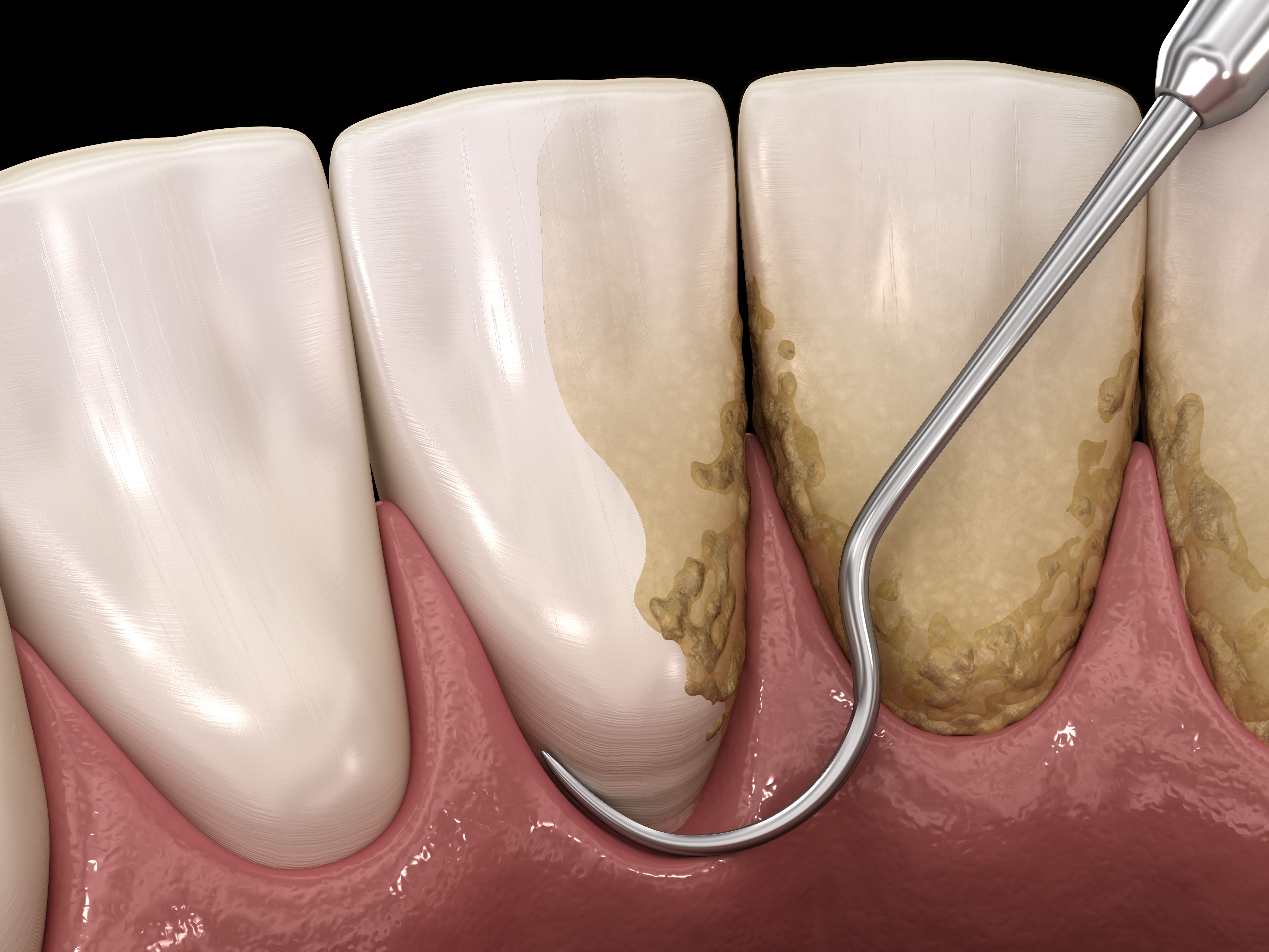
Adjunctive Therapeutic Agents: Systemic and Locally Delivered Antimicrobials
Periodontitis is understood to have a multifactorial origin that results from a complex interaction between the host’s immune system and dysbiotic plaque biofilms. Before scientists were able to comprehend biofilm presence and its role in chronic inflammation, a commonly accepted theory (specific plaque hypothesis by Loesche) advocated diagnosing and treating periodontitis based on prominent bacterial types.1 Nonsurgical periodontal therapy for the “gums” became increasingly popular with the public, and it was a movement that started in the late 1970s by Dr Paul H. Keyes, even though it wasn’t scientifically tested at the time. Microscopic examination of plaque with a phase contrast microscope, scaling and root planing (SRP), and assiduous daily home care involving antibacterial agents like baking soda and hydrogen peroxide and systemic antibiotics were part of the Keyes technique.2
Over the last 4 to 5 decades, clinicians have used various local or systemic agents including host modulation agents. In assessing the effects of systemic antimicrobials as an adjunct to SRP for nonsurgical periodontal treatment, the Cochrane Oral Health Group published a systematic review (SR) in 2020.3 It reported on a range of antibiotics with different dosages and combinations. No serious adverse effects from the various combinations of systemic antibiotics were reported, but there were adverse events such as nausea, vomiting, diarrhea, mild gastrointestinal disturbances, and metallic taste. Unfortunately, there is insufficient evidence to decide whether 1 combination of antibiotics is better than another and there is very low certainty of evidence (for long-term follow-up) to inform clinicians and patients if adjunctive antimicrobials are of any help in the nonsurgical treatment of periodontitis.3
In 2015, The American Dental Association (ADA) Council on Scientific Affairs convened a panel of experts to formulate clinical practice guidelines on nonsurgical treatments of patients with periodontitis by means of SRP with and without adjuncts.4 The clinical practice guideline is based on a SR of 72 research articles providing clinical attachment levels on clinical trials of 6-month duration and published through July 2014. SRP showed a moderate benefit for patients with periodontitis. Systemic subantimicrobial-dose doxycycline and systemic antimicrobials showed similar magnitudes of benefits as adjunctive therapies to SRP but the systemic antimicrobials were recommended at different strengths. The panel recommended systemic subantimicrobial-dose doxycycline because of the higher potential of adverse effects with higher doses of systemic antimicrobials. Other adjuncts that were evaluated and were labeled weak included chlorhexidine chips and photodynamic therapy with a diode laser. Evidence was lacking and level of certainty of evidence was low for doxycycline hyclate gel and minocycline microspheres and other lasers as SRP adjuncts.4
In summary, none of the systemic antimicrobials or local delivery antimicrobials have high-certainty evidence to support their use as adjuncts to SRP with the exception of subantimicrobial dose doxycycline.
Perioscopy or Periodontal Endoscopy
The primary objective of SRP is to restore periodontal health by removing pathogenic materials and products (eg, biofilm, calculus, and endotoxins) that induce inflammation from periodontally involved root surfaces. Calculus has been shown to contain bacterial products that contribute to the inflammatory response and can perpetuate periodontal infection.5-7 Numerous studies, however, indicate clinical difficulty in determining the thoroughness of subgingival instrumentation as well as clinical limitations in instrumenting subgingival root surfaces in a closed pocket, particularly in pocket depths greater than 5 mm.8-11
The periodontal endoscope facilitates visualization of the subgingival environment via a 1 mmdiameter, flexible, fiber-optic probe with real-time video imaging capabilities. This fiber-optic system enlarges the image 20 to 40 times while delivering light and irrigation to the working field.
An early study by Stambaugh et al evaluated the periodontal endoscope and the ability of the clinician to develop skills in using fiber optics to accurately visualize the contents of the subgingival sulcus.12 Results from other studies demonstrated that adjunctive use of the periodontal endoscope for nonsurgical periodontal therapy resulted in improved visibility of deposits for calculus removal.13
Wilson et al found that root surfaces treated endoscopically with a single course of closed subgingival SRP demonstrated no histologic signs of chronic inflammation 6 months after therapy.14
The 2015 ADA Center for Evidence-Based Dentistry clinical practice guidelines on the nonsurgical treatment of chronic periodontal disease note that SRP IS still the gold standard for non-surgical therapy.4 The periodontal endoscope supports this therapeutic approach and has been found to be a minimally invasive technology that provides visualization with magnification and illumination within a closed pocket to aid in the diagnosis and treatment of chronic periodontitis and peri-implant disease.
References
- Loesche WJ. Clinical and microbiological aspects of chemotherapeutic agents used according to the specific plaque hypothesis. J Dent Res. 1979;58(12);2404-2412.
- Canadian Academy of Periodontology. CAP position on Keyes Technique. Accessed March 2022. https://www.cap-acp.ca/en/public/keyes.html
- Khattri S, Kumbargere Nagraj S, Arora A, et al. Adjunctive systemic antimicrobials for the non‐surgical treatment of periodontitis. Cochrane Database Syst Rev. 2020;11:CD012568.
- Smiley CJ, Tracy SL, Abt E, et al. Evidence-based clinical practice guideline on the nonsurgical treatment of chronic periodontitis with and without adjuncts. J Am Dent Assoc. 2015;146(7):525-535.
- Allen DL, Kerr DA. Tissue response in the guinea pig to sterile and non-sterile calculus. J Periodontol. 1965;36:121-126.
- Tan BKT, Mordan NJ, Embleton J, Pratten J, Galgut PN. Study of bacterial viability within the human supragingival dental calculus. J Periodontol. 2004;75(1):23-29.
- Blue CM, Lenton P, Lunos S, Poppe K, Osborn J. A pilot study comparing the outcome of scaling/root planing with and without Perioscope technology. J Dent Hyg. 2013;87(3):152-157.
- Rabbani GM, Ash MM, Caffesse RG. The effectiveness of subgingival scaling and root planing in calculus removal. J Periodontal. 1981;52(3):119-123.
- Caffesse RG, Sweeney PL, Smith BA. Scaling and root planing with and without periodontal flap surgery. J Clin Periodontol. 1986;13(3):205-210.
- Sherman PR, Hutchens LH Jr., Jewson LG, Moriarty JM, Greco GW, McFall WT Jr. The effectiveness of subgingival scaling and root planing. I. Clinical detection of residual calculus. J Periodontol. 1990;61(1) 9-15.
- Stambaugh RV, Dragoo M, Smith DM, Carasali L. The limits of subgingival scaling. Int J Periodontics Restorative Dent. 1981;1(5):30-41.
- Stambaugh RV, Myers G, Ebling W, Beckman B, Stambaugh K. Endoscopic visualization of the submarginal gingiva dental sulcus and tooth root surfaces. J Periodontol. 2002;73(4):374-382.
- Osborn JB. Role of the dental endoscope in calculus detection. Dimens Dent Hyg. 2016;14(2):40,42-44.
- Wilson TG Jr, Carnio J, Schenk R, Myers G. Absence of histologic signs of chronic inflammation following closed subgingival scaling and root planning using the dental endoscope: human biopsies — a pilot study. J Periodontol. 2008;79(11):2036–2041.