Surround Medical Systems Announces FDA Pre-Market Clearance for PORTRAY Dental Imaging System
This new imaging system that utilizes 2D and 3D technologies has received the pre-clearance necessary to go on the market for dental practices.
Surround Medical Systems
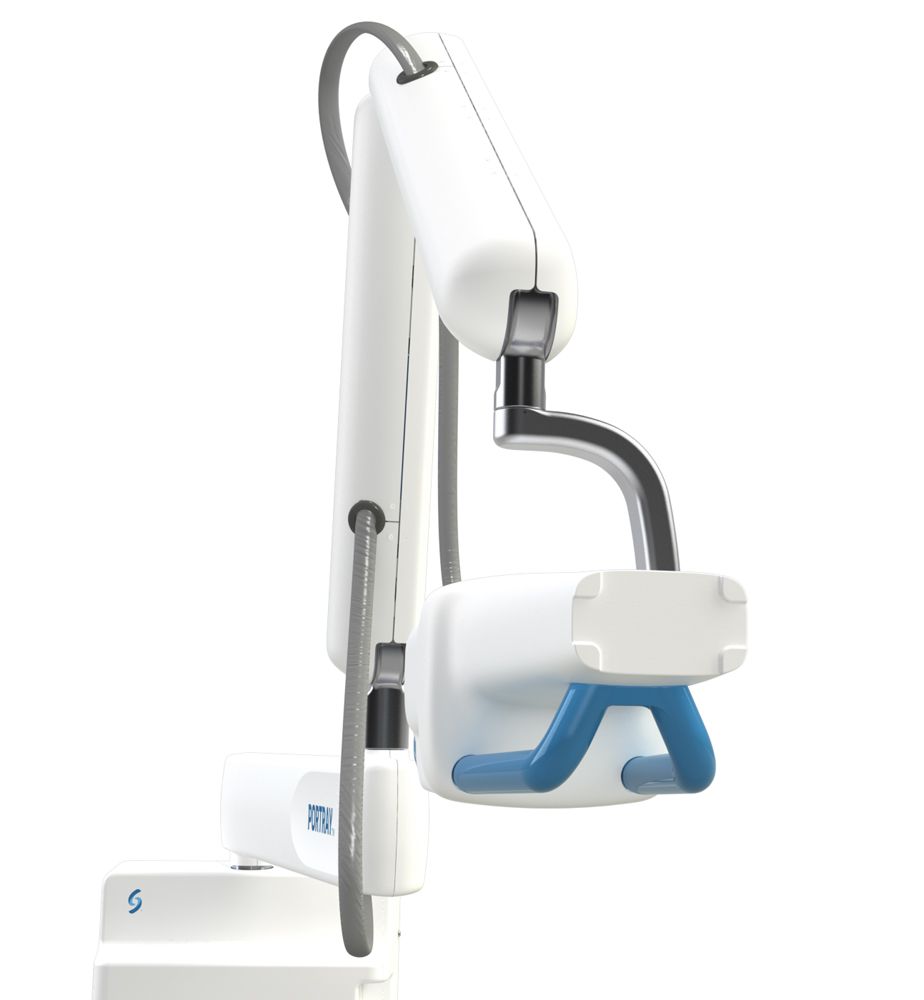
Surround Medical Systems, Inc. has announced that its PORTRAY™ dental imaging system has received 510(k) pre-market clearance from the United States Food and Drug Administration (FDA).
PORTRAY™ is a 2D diagnostic tool that clinicians can use to obtain 3D tomosynthetic and 2D synthetic imaging for producing diagnostic dental radiographs. The PORTRAY™ system is said to be able to detect decay earlier than standard X-ray systems in its deployment of multiple beams. These multiple beams will create image “slices” which can then be reconstructed to create 3D images.
President and Chief Executive Office of Surround, David LaVance, said in a press release, that this system is a leap forward for dental imaging.
“The PORTRAY™ system provides dentists with an intraoral 3D dental imaging system that will allow the dentists to provide a higher level of care for patients by detecting cavities and fractures that would otherwise go undetected by current 2D intraoral dental imaging systems,” LaVance said in the press release. “The PORTRAY™ system has the potential to substantially change and improve dental techniques and provide patients better outcomes and improved health.”
The technology for the PORTRAY™ system comes from research done at the University of North Carolina at Chapel Hill. This research combined the works of the Department of Physics and Astronomy as well as the Adams School of Dentistry. With this FDA pre-clearance, Surround expects the PORTRAY™ system to be available for sale for dental practices in late 2021.