Sunstar Americas Issues Voluntary Nationwide Recall of GUM Paroex Oral Rinse Due to Microbial Contamination
Company’s chlorhexidine gluconate oral rinse USP, 0.12% products bearing an expiration date from 6/30/22 to 9/30/22 are being recalled.

Sunstar Americas, Inc., announced Tuesday that is voluntarily recalling Paroex® Chlorhexidine Gluconate Oral Rinse USP, 0.12% products bearing an expiration date from 6/30/22 to 9/30/22 to the consumer level. The recall was issued because this product may be contaminated with the bacteria Burkholderia lata.
Use of the defective product in the immunocompetent host may result in oral and, potentially, systemic infections requiring antibacterial therapy, according to Sunstar Americas. In the most at-risk populations, the use of the defective product (see specific lots below) may result in life-threatening infections, such as pneumonia and bacteremia. To date, no adverse events have been reported to the company related to this recall.
The prescription oral rinse product, available through healthcare professionals only, is indicated for use as part of a professional program for the treatment of gingivitis and is packaged as follows:
1789P GUM® Paroex® is distributed in cases each containing 6 amber bottles of 16 fluid ounce (473 ml) chlorhexidine rinse. The bottle has a childproof cap and a 15 ml metered dosage cup, is safety sealed, and is decorated with a multiple-panel wrap-around label. 1788P GUM Paroex is distributed in cases each containing 24 amber bottles of 4 fluid ounce (118.25 ml) chlorhexidine rinse. The bottle for this product also has a childproof cap, is safety sealed, and is decorated with a multiple-panel wrap-around label.

Paroex was distributed nationwide to dental offices, dental distributors, pharmaceutical wholesalers, dental schools, and pharmacies, Sunstar America states.
The company is notifying its direct distributors and customers by USPS Priority mail and is arranging for return of all recalled products. Patients, pharmacies, and healthcare facilities in possession of these products should stop using and dispensing immediately.
Consumers with questions regarding this recall can contact Sunstar Americas by phone at 800-528-8537 or email us.pcr@us.sunstar.com on Monday-Friday from 8am-5pm CST. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to using this drug product.
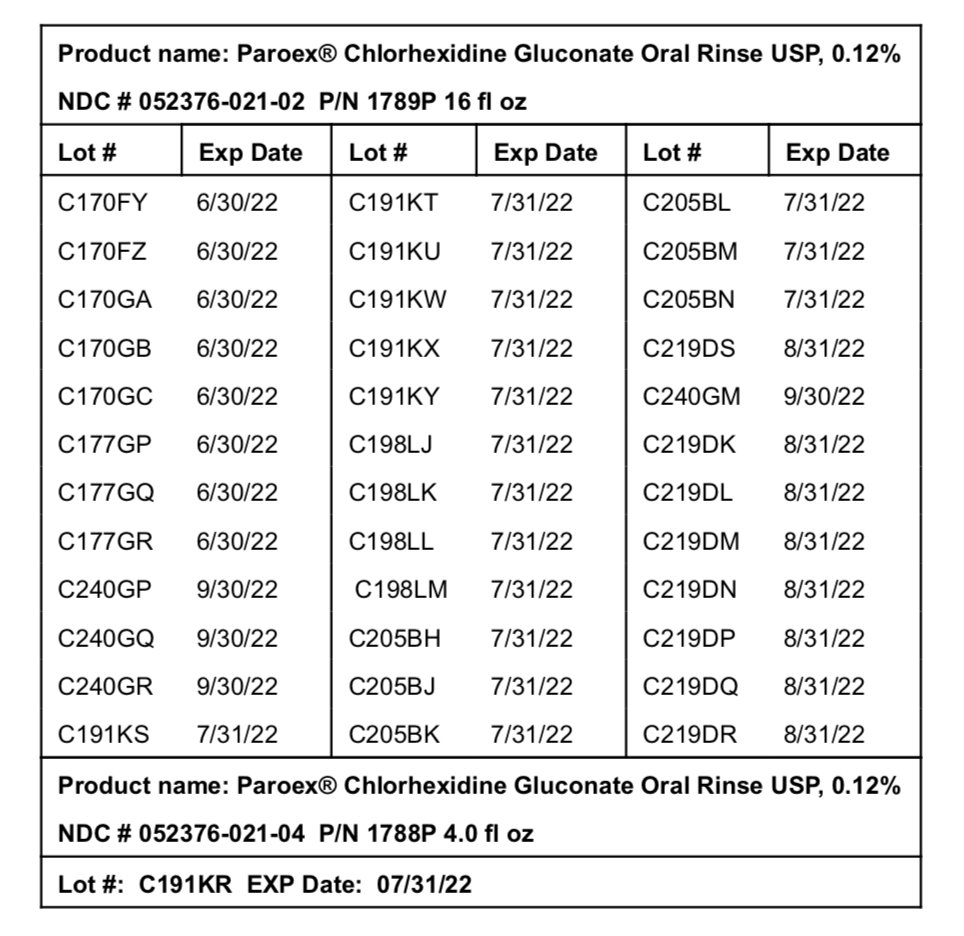
Affected products and lot numbers follow below:
AFFECTED LOTS
Product name: Paroex® Chlorhexidine Gluconate Oral Rinse USP, 0.12% NDC # 052376-021-02 P/N 1789P 16 fl oz
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
Complete and submit the report Online: www.fda.gov/medwatch/report.htm
Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 800-FDA-0178.
Sunstar is committed to delivering safe, fully compliant products of the highest quality and is taking necessary steps to prevent future occurrence of this issue, the company states in Tuesday’s press release.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.