Solvay Dental 360 Announces Partnership with Amann Girrbach
Ultaire AKP high-performance polymer for removable partial denture frames now available for market-leading CAD/CAM system.
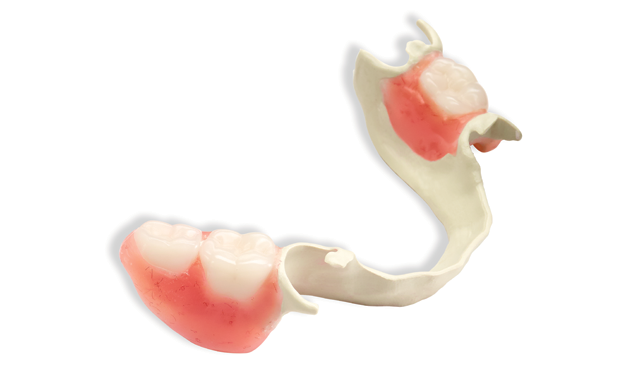
Solvay Dental 360™, today announced that Ultaire™ AKP, a high-performance polymer for removable partial denture (RPD) frames, is now validated for milling in Ceramill CAD/CAM workflows through Amann Girrbach.
“We are pleased to partner with an innovative industry leader like Amann Girrbach to offer Ultaire™ AKP in the digital workflow of the Ceramill CAD/CAD system,” says Shawn Shorrock, global director, Solvay Dental 360™. “Our partnership will make removable partial dentures made from Ultaire™ AKP even more accessible to the labs, dentists and patients who need them, streamlining the fabrication process and improving the patient experience.”
Ultaire™ AKP is an aryl ketone polymer that provides a comfortable, lightweight and more aesthetically pleasing alternative to traditional metal RPD frames. Ultaire™ AKP is a rigid material - not flexible - suitable as a metal replacement and different than the other non-metal materials currently on the market. It is both strong enough and tough enough to be tooth-supported and help reduce bone loss, yet flexible enough to engage undercuts more deeply.

The manufacturing process for RPDs made with Ultaire™ AKP is now validated and automatically stored in the Ceramill software with all the necessary design parameters and milling strategies. This considerably simplifies the training and qualification process for Amann Girrbach users and can be carried out directly by Amann Girrbach's Helpdesk in just a few steps. Ultaire™ AKP can also be obtained directly from Amann Girrbach and its dealers in Germany, or through Solvay Dental 360™ in North America and the U.K. The latest version of the software upgrade module Ceramill M-Part for RPDs and the Ultaire™ AKP module will be available following the Ceramill Suite 3.9 update in August 2018.
“We are proud to offer the innovative Ultaire™ AKP high-performance polymer for RPDs to our customers through a sophisticated and consistent digital workflow in our Ceramill CAD/CAM system,” said Christian Ermer, Amann Girrbach head of product management. “Ultaire™ AKP design parameters differ from those of metal, so our software incorporates customized CAD strategies that are essential for prosthetic success. In addition, our specially designed CAM milling strategies guarantee the quality and precision of the final product.”
Solvay Dental 360™ is a new dental business and medical device line of Belgian multi-specialty chemical group Solvay, a global leader in high-performance polymers used in the healthcare industry for medical and dental devices, equipment and instrumentation, including implantable applications. With over 35 brands available in more than 1,500 formulations, Solvay has the broadest portfolio of high-performance healthcare polymers in the world.
The Solvay Dental 360™ business line was developed by Solvay’s Specialty Polymers Global Business Unit. Through products like Ultaire™ AKP, and other innovations to be commercialized in the future, Solvay Dental 360™ is committed to developing new and innovative materials that improve patient outcomes and advance the dental industry.
Ultaire™ AKP is available in North America, U.K. and Germany and has regulatory approval 510K clearance, CMDCAS and EU. It is made in the U.S. under FDA and International Organization for Standardization (ISO) guidelines.
Additional information about the Ultaire™ AKP digital workflow in the Ceramill CAD/CAM system through Amann Girrbach is available here. For more information about Ultaire™ AKP or Solvay Dental 360™, visit https://www.solvaydental360.com/.