New Nasal Spray Deemed Safe and Effective Anesthesia for Dental Procedures
A successful Phase 3 trial suggests dentists may soon be able to use a nasal spray instead of a syringe when numbing patients for procedures.
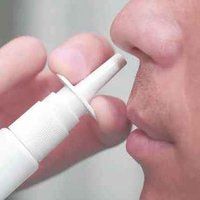
A new nasal spray recently approved by the Food and Drug Administration (FDA) has proved to be safe and effective in a Phase 3 clinical trial led by a
University of Pennsylvania School of Dental Medicine
research team. The nasal spray is the first of its kind to be used in dentistry as an alternative to the injection of a numbing agent prior to procedures.
The nasal spray,
, has been in development for over 10 years. The spray combines tetracaine HCl, a local anesthetic, and oxymetazoline HCl, a nasal decongestant, in a single spray dose. It is intended for use as a topical anesthetic for restorative procedures on teeth 4-13 and A-J in adults and also in children who weigh 40 kg or more.
The research was conducted during the Phase 3 double-blind, randomized clinical trial with 150 adult participants, 100 of whom received the actual product instead of a placebo. Kovanaze was used as an anesthetic during single dental filling procedures involving the upper bicuspids, canines, or incisors. Each patient received one spray, waited four minutes, and then received a second spray. Ten minutes after receiving the second spray, test drilling was performed to determine whether the anesthetic was effective. If not, the patient received a third spray of the product. If, after three sprays, patients still experienced pain, an injection of a common local anesthetic was provided in order to complete the procedure.
In 88% of participating patients, pain was completely prevented by Kovanaze. This success rate is comparable to commonly used injectable anesthetic agents. Patients reported runny nose and nasal congestion as the most common side effects. No serious side effects were reported.
As a follow up to the trial, researchers plan to test whether Kovanaze could be used in more invasive dental procedures, like root canals or oral tissue biopsies. Further studies should also evaluate whether the product can be used in children weighing less than 40 kg.
Many individuals fear the pain that might come with certain dental treatments. As a result, they often delay or avoid treatment altogether. Researchers hope that Kovanaze could replace the need for injections of anesthetics, which are sometimes the most painful part of dental procedures. As Dr. Elliot Hersh, the trial’s lead author, noted, "there is really nothing else like this. It would certainly make for a more stress-free dental office visit for children as well as adults if we could replace some of these anesthetic injections with a simple spray.”