Innovator Profile - New implant solutions from DENTSPLY Implants
Dental implantology remains a major growth area in all levels of the dental workflow. DENTSPLY Implants continues to be a major factor in the implants market, releasing solutions, support and innovations that are pushing the industry forward.
Talking about new dental implant designs, digital workflows and how labs can prepare for the future of implantology.
Dental implantology remains a major growth area in all levels of the dental workflow. DENTSPLY Implants continues to be a major factor in the implants market, releasing solutions, support and innovations that are pushing the industry forward.

We recently spoke with DENTSPLY Implants’ Matt Gassel, director of marketing, North America, and Mikael Sander, group vice president, digital implant solutions, about the new implant design and how DENTSPLY Implants will support labs into the future.
This article originally appeared in the July/August issue of Dental Lab Products. For more great articles about what's happening in the dental lab world, click the issue below:
Related: New treatment solutions from DENTSPLY Implants
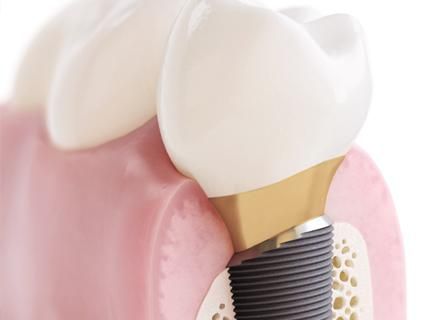
DLP: You’ve just introduced the OsseoSpeed Profile EV. How is that helping dentists, labs and patients?
A: As a part of the ASTRA TECH Implant System EV, recognized for its foundation of science and documented marginal bone maintenance, the introduction of the OsseoSpeed Profile EV takes that concept of clinical versatility and ease of use even further as an implant uniquely designed especially for sloped ridge situations. As with all OsseoSpeed EV implants, the self-engaging design of the impression components allow impression taking to be remarkably easier. The entire OsseoSpeed EV implant line, including OsseoSpeed Profile EV is designed for one-position-only placement of ATLANTIS patient-specific abutments, which makes it extremely user-friendly for both laboratories and restorative doctors to integrate ATLANTIS into their businesses and collaborate in delivering implant-supported restorations with the highest level of form, function and esthetics for the patient.
Q: How is the increasing popularity of digital workflows helping DENTSPLY Implants and its customers?
A: The increasing prevalence of a digital workflow allows us to connect with our customers in many more ways that increase the efficiency and accuracy of the implant treatment process for improved time- and cost-savings and overall treatment outcomes. In fact, within our comprehensive portfolio supporting all phases of implant therapy, customers are able to leverage a digital workflow beginning with treatment planning (SIMPLANT) through to the final restoration (ATLANTIS).
The ATLANTIS solution accepts both intraoral and lab-based scans to provide the data needed for the design and manufacture of a patient-specific abutment or suprastructure. Innovative digital products such as the ATLANTIS Abutment Core File, which is a digital representation of the outer surface of an ATLANTIS™ Abutment that enables the design of a precise coping and final restoration even before the physical abutment is received, save our laboratory customers time and labor.
Related: 3Shape adds DENTSPLY Implants to their Implant Module
Most all our digital solutions are “open platform,” which means they are compatible for all major implant systems and scanners so our clinical and laboratory customers have full flexibility in working with their systems of choice and equipment they have already invested in.
Q: How does DENTSPLY Implants make sure its solutions are connected to existing workflows used by labs and dentists?
A: We are constantly striving to be the easiest company to work with in the digital space, and we also want to ensure our solutions are well-tested and work well when we bring them to the market. We are consistently adding to our list of compatible sources and have developed both dedicated workflows for specific scanners as well as open STL solutions to reach as many customers as possible. We want our customers to use the equipment they have already invested in and not have to purchase special dedicated equipment. Finally, we keep a close eye on market developments and work to develop partnerships with the leading companies so we can better serve our customers.
Q: Where do you see the implant world going in the next three years? .
A: We believe implant therapy will continue to grow at a good pace and digital collaboration between treatment team members will become much more common than it is today. There will be more digital planning based on CBCT data, and the prosthetics will become more integrated into that process than ever before. Because each patient is a unique individual, digital workflows and advancements in technology will also allow for increased simplicity and precision in providing truly patient-specific solutions.
Q: How does DENTSPLY Implants help navigate those changes?
A: At DENTSPLY Implants, we respect the team approach to implant therapy and believe that helping to provide solutions and services that help enhance the coordination and communication among team members is critical. The dental laboratory is a key member of that team, and, for this reason, we have field representatives who are dedicated to supporting our laboratory customers so they can be part of leading the trend with their clinical partners.
Q: In your own words, why do you think labs should choose DENTSPLY Implants as a partner?
A: There are many factors that laboratories and clinicians need to consider when choosing who to work with and who to purchase their implant products and services from. At DENTSPLY Implants, we do not want to be just a supplier. Through our products and education, we want to be a true partner with dental laboratories and clinicians in helping them deliver consistent and high-quality implant dentistry. We also want to help them grow this portion of their business to their desired goal.
How Abutment Core File from DENTSPLY Implants makes abutment placement easier
Our vision is a world where everyone eats, speaks and smiles with confidence. To make that vision a reality, we hope to be the number one choice for laboratories and clinicians by continuing to provide well-documented and innovative solutions that meet dental professionals’ needs and patients’ demands.
Editor's note: Innovator Profiles are part of a special advertising section in the pages of Dental Lab Products and are paid for by advertising partners. The opinions in these profiles are not necessarily reflected by Dental Lab Products, its editorial team or UBM Medica.