3D Systems NextDent Denture 3D+ cleared by FDA
The new biocompatible denture material received 510(k) clearance from the US Food and Drug Administration on Nov. 1.
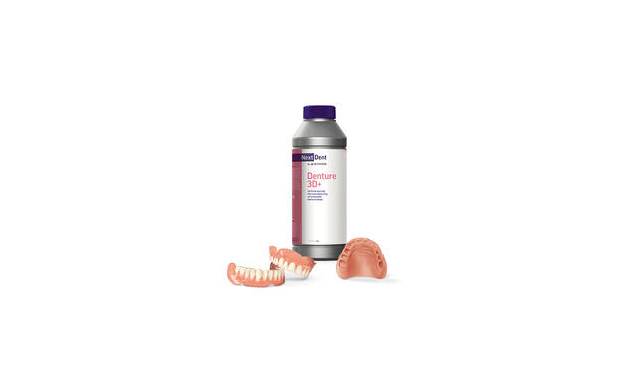
3D Systems announced FDA-clearance for its NextDent® 3D+ line of dental materials, the company said in a press release issued today.
The new biocompatible denture material received 510(k) clearance from the US Food and Drug Administration on Nov. 1. The combination of this new dental 3D printing material, NextDent 5100 3D printer, and intraoral scanning and dental software solutions offers end-to-end digital dentistry solutions, the company said.
NextDent Denture 3D+ is a biocompatible Class IIa material designed for printing all types of removable denture bases. It is available in five new colors-dark pink, light pink, opaque pink, red pink, and translucent pink.
Producing a denture using NextDent Denture 3D+ for the base, and NextDent C&B MFH for the teeth on the NextDent 5100 results in a final denture that is 90 percent less expensive to produce in 75 percent less time than dentures created using analog methods, according to the company’s statement.
“With 3D Systems’ Digital Denture Workflow, dental laboratories and clinics are now able to
produce dental devices at dramatically increased speed while reducing material waste and capital equipment expenditure as well as reliance upon milling centers,” said Rik Jacobs, 3D Systems’ vice president, general manager, dental. “FDA clearance of NextDent Denture 3D+ is the last piece that creates a trusted end-to-end workflow – giving prosthodontists a competitive advantage while improving the patient experience.”
NextDent Denture 3D+ material reportedly has excellent mechanical properties capable of producing dentures that rival conventional denture base materials. 3D Systems also offers one of the largest color palettes available for denture base material, allowing labs and clinics to deliver a final product with a natural-looking aesthetic.
3D Systems is an innovator in the 3D printing manufacturing industry. For more information, visit 3dsystems.com.